LCD substrate glass enables TFT-LCDs and requires ultra-pure, alkali-free, thermally stable glass. Complex processes and global oligopolies dominate production due to high technical barriers.
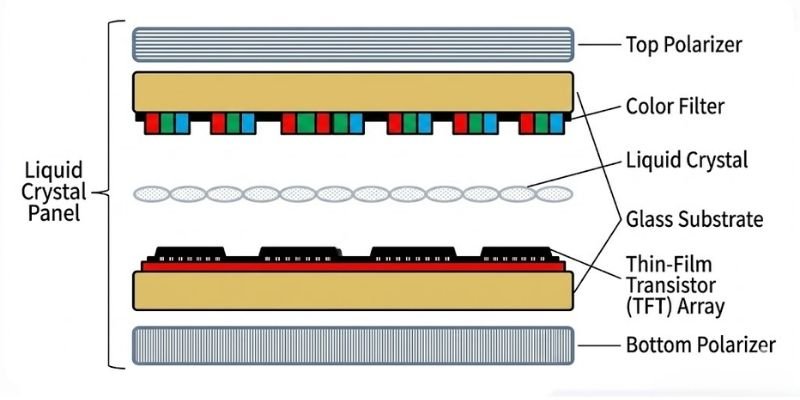
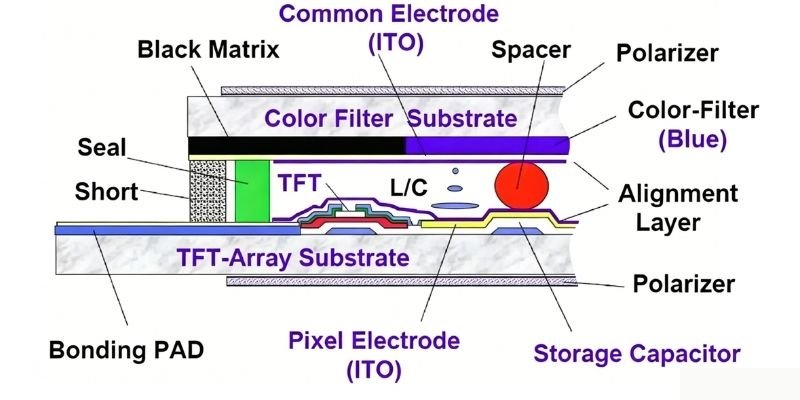
Substrate glass is one of the most important raw materials used in LCD panels. The key structure of an LCD panel resembles a sandwich: two layers of “bread” (the TFT substrate and color filter) enclose the “filling” (liquid crystal). Therefore, manufacturing a TFT-LCD panel requires two pieces of glass — one serving as the bottom glass substrate and the other as the color filter substrate.
Substrate glass accounts for approximately 20% of the raw material cost of a TFT-LCD panel and has a significant impact on product performance. Key panel characteristics such as resolution, light transmittance, thickness, weight, and viewing angle are all closely related to the quality of the substrate glass used. As a critical foundational material, substrate glass plays a role in the TFT-LCD industry comparable to silicon wafers in the semiconductor industry.
TFT-LCD Panel Structure
Substrate glass is one of the most important raw materials used in LCD panels. The key structure of an LCD panel resembles a sandwich: two layers of “bread” (the TFT substrate and color filter) enclose the “filling” (liquid crystal). Therefore, manufacturing a TFT-LCD panel requires two pieces of glass — one serving as the bottom glass substrate and the other as the color filter substrate.
Substrate glass accounts for approximately 20% of the raw material cost of a TFT-LCD panel and has a significant impact on product performance. Key panel characteristics such as resolution, light transmittance, thickness, weight, and viewing angle are all closely related to the quality of the substrate glass used. As a critical foundational material, substrate glass plays a role in the TFT-LCD industry comparable to silicon wafers in the semiconductor industry.
Types of Substrate Glass
Substrate glass can generally be divided into two categories: alkali-containing glass and alkali-free glass.
Alkali-containing glass is mainly used in TN/STN LCD panels. However, for TFT-LCD applications, alkali metal ions inside the glass can affect the stability of thin-film transistor gate voltage. Therefore, TFT-LCD manufacturing requires alkali-free glass formulations that exclude sodium oxide and potassium oxide.
While sodium oxide and potassium oxide help lower the melting temperature of glass, alkali-free glass requires significantly higher furnace temperatures during production. This is one of the reasons why alkali-free glass manufacturing is technically more challenging than conventional alkali glass production.
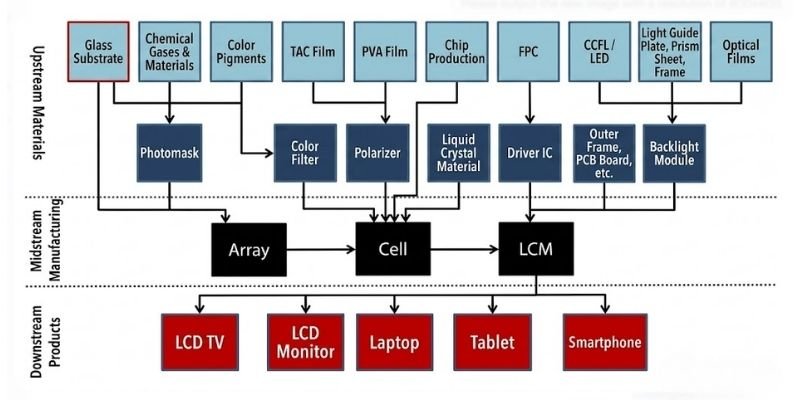
TFT-LCD Industry Chain
As one of the foundational raw materials for LCD panels, substrate glass occupies the upstream end of the LCD industry chain.
Its upstream raw materials include basic chemical materials such as quartz powder and alumina. Downstream customers mainly consist of panel manufacturers and color filter suppliers, which use the glass to manufacture TFT arrays and color filters before liquid crystal injection and module assembly are completed to form Open Cell panels.
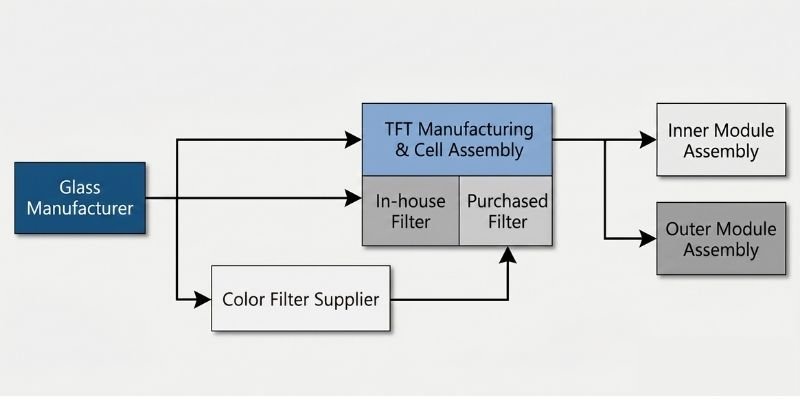
Substrate Glass Supply Chain
As the cornerstone of the thin-film display industry, substrate glass is widely used not only in TN/STN and TFT LCD structures, but also serves as an essential substrate material for OLED displays. The importance of substrate glass is largely unaffected by changes in display technology mechanisms, making it an irreplaceable material with a stable long-term industry position.
Characteristics of Substrate Glass
Chemical Properties
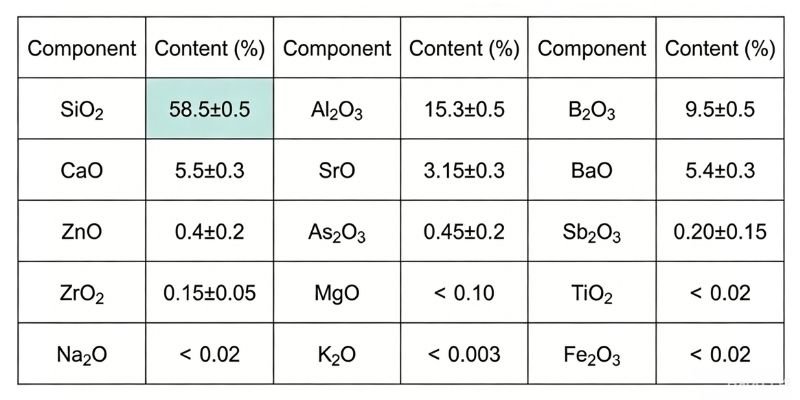
Main Physical and Chemical Parameters
(1) Strain Point
During TFT manufacturing processes, substrate glass undergoes repeated thermal treatments at temperatures up to 625°C. The substrate must remain rigid at these temperatures without exhibiting viscous flow behavior. Otherwise, deformation and thermal stress can occur during cooling, resulting in dimensional changes.
Therefore, the strain point of substrate glass must exceed 625°C. Including a safety margin of 25°C, the strain point should generally be at least 650°C.
(2) Chemical Stability
Substrate glass must withstand various harsh chemical treatments used in display manufacturing processes.
For example, α-Si active matrix LCD manufacturing involves more than seven layers of thin-film circuitry and multiple etching processes. Corrosive chemicals and cleaning agents range from strong acids to strong alkalis, including:
- 10%+ NaOH
- 10%+ H₂SO₄
- Concentrated HNO₃
- 10% HF-HNO₃
- Concentrated H₃PO₄
As a result, substrate glass has some of the strictest chemical stability requirements among all glass products.
(3) Alkali Restrictions
Because electronic circuits are printed directly onto the substrate glass, the glass must contain extremely low levels of monovalent alkali metals (R₂O), ideally approaching zero.
If monovalent alkali metals are present, they may migrate from inside the glass to the surface at high temperatures, potentially causing short circuits or circuit failures.
The monovalent alkali metals referenced here mainly include:
- Potassium (K⁺)
- Sodium (Na⁺)
- Lithium (Li⁺)
(4) Thermal Expansion Coefficient
During LCD manufacturing, silicon layers are deposited onto the glass surface. Therefore, the thermal expansion coefficient of the glass must closely match that of silicon.
Silicon dioxide typically has a thermal expansion coefficient of approximately:
5–7 × 10⁻⁶ /°C
In addition, displays undergo repeated rapid heating and cooling cycles during manufacturing. These thermal cycles can relax the glass structure and cause dimensional changes, potentially resulting in photolithography alignment errors.
Therefore, extremely low thermal shrinkage is required, often limited to only a few micrometers.
Generally, the thermal expansion coefficient of substrate glass should remain below:
40 × 10⁻⁷ /°C within the 0–300°C temperature range.
Glass Raw Materials and Characteristics
The primary raw materials used in LCD substrate glass include:
- Quartz powder
- Strontium carbonate
- Barium carbonate
- Boric acid
- Boron oxide
- Alumina
- Calcium carbonate
- Barium nitrate
- Magnesium oxide
- Tin oxide
- Zinc oxide
These materials function as glass formers, modifiers, and intermediate components, collectively determining the physical and chemical properties of the final glass.
Quartz Powder
The primary component of quartz powder is SiO₂, the main oxide responsible for glass formation.
SiO₂ forms an irregular continuous network based on the silicon-oxygen tetrahedral structure [SiO₄], which serves as the backbone of the glass structure.
SiO₂ can:
- Lower thermal expansion coefficient
- Reduce density
- Increase strain point
Insufficient SiO₂ content can reduce acid resistance and overall chemical stability while making it difficult to achieve low expansion, low density, and high strain-point glass.
Alumina
Alumina (Al₂O₃) is an intermediate oxide.
When oxygen is insufficient, aluminum forms [AlO₆] octahedra in the network gaps. When excess oxygen exists, aluminum forms [AlO₄] tetrahedra within the glass network, strengthening the structure.
Al₂O₃ can:
- Improve glass stability
- Lower thermal expansion coefficient
- Increase strain point
- Improve elastic modulus
- Enhance chemical stability
The combined content of silicon oxide and alumina generally exceeds 70% of the total raw material composition.
Boric Acid (Boron Oxide)
Boric acid acts as a fluxing agent.
B₂O₃ reduces glass melting viscosity without significantly increasing thermal expansion. Appropriate amounts improve hydrofluoric acid resistance and processability.
However, excessive B₂O₃:
- Reduces acid resistance
- Lowers strain point
- Reduces transition temperature
- Harms chemical durability
For this reason, boron oxide content is typically maintained below 10%.
Because boron oxide is highly sensitive to moisture during production, humidity control during storage is critical.
Strontium Carbonate
Strontium carbonate primarily introduces strontium oxide (SrO).
SrO can:
- Improve melting characteristics
- Avoid excessive density increases
- Prevent significant increases in thermal expansion coefficient
- Limit strain point reduction
However, excessive SrO may negatively affect acid resistance and durability against alkaline stripping agents.
SrO also provides X-ray absorption properties.
Zinc Oxide
ZnO is an intermediate oxide.
Under suitable oxygen conditions, ZnO can form [ZnO₄] tetrahedra within the glass network, improving structural stability.
ZnO can:
- Lower thermal expansion coefficient
- Improve chemical stability
- Enhance thermal stability
- Increase refractive index
Tin Oxide
Tin oxide is used as a refining agent to replace toxic arsenic oxide in glass manufacturing.
Mainstream Manufacturing Processes for Substrate Glass
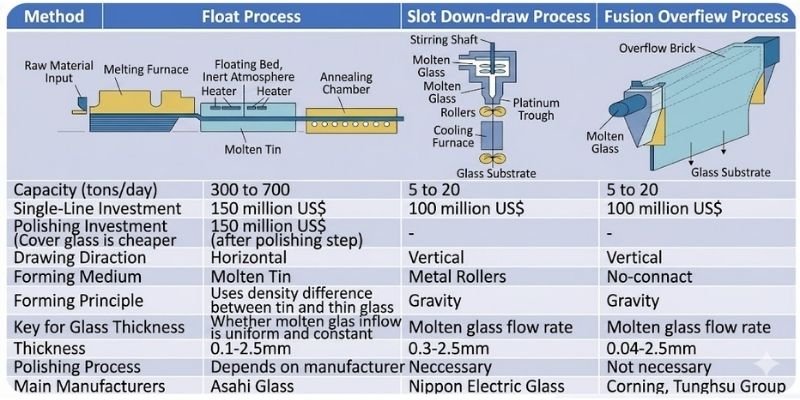
The three primary manufacturing processes for substrate glass are:
- Float Process
- Slot Down-Draw Process
- Overflow Fusion Process
Currently, the overflow fusion process is the dominant mainstream technology.
Float Process
The float process is the most widely used and historically established flat glass manufacturing technology.
Molten glass is transferred onto a bath of molten tin. Due to density differences between tin and glass, the molten glass naturally spreads flat under surface tension and gravity before entering the cooling chamber.
Float glass requires additional downstream grinding and polishing processes.
Advantages include:
- High production capacity
- Easy scalability for larger substrate sizes
- Lower manufacturing cost
However, post-processing partially offsets these cost advantages.
Float technology was initially used mainly for TN/STN substrate glass. Later, AGC successfully adapted float technology for alkali-free TFT substrate glass manufacturing.
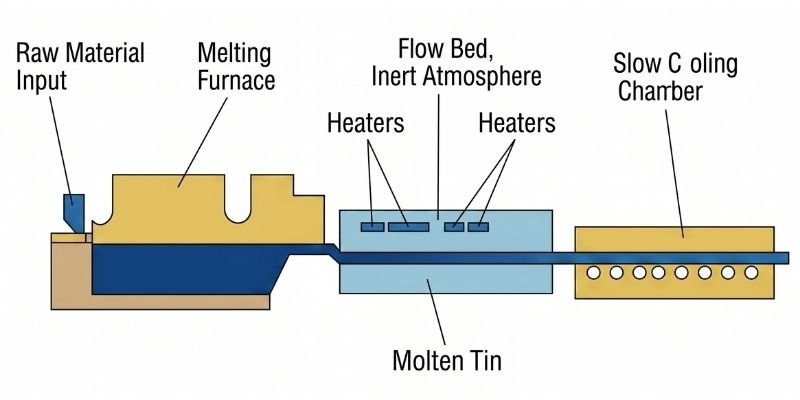
Slot Down-Draw Process
In the slot down-draw process, molten glass flows through platinum alloy slots under gravity and is shaped through rollers before cooling and solidification.
The size of the slots and draw speed determine glass thickness, while temperature distribution affects flatness.
However:
- Slot deformation may affect yield rates
- Roller contact impacts surface flatness
- Additional polishing is required
This process was primarily used by Nippon Electric Glass but has gradually been phased out due to limited advantages.
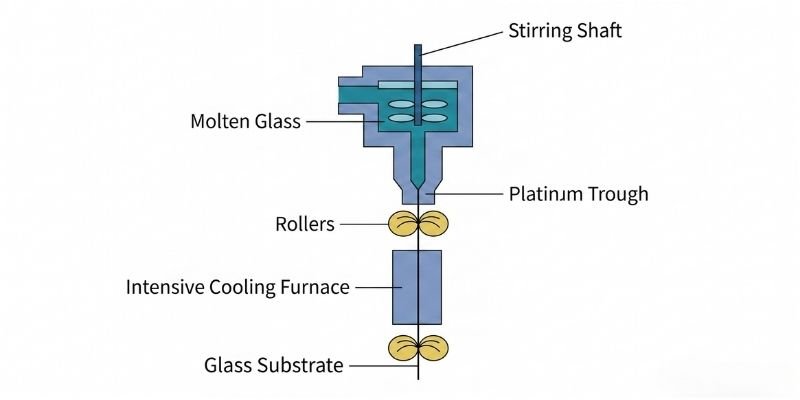
Overflow Fusion Process
In the overflow fusion process, molten glass flows into a trough and overflows symmetrically down both sides, similar to a waterfall, before merging into a continuous glass sheet.
This process was pioneered by Corning.
Because the glass does not contact external media during forming — unlike molten tin in float processing or rollers in down-draw methods — surface quality remains extremely uniform.
As a result:
- No polishing is required
- Surface contamination risks are minimized
- Surface flatness is significantly improved
Today, overflow fusion has become the mainstream manufacturing technology for TFT-LCD substrate glass.
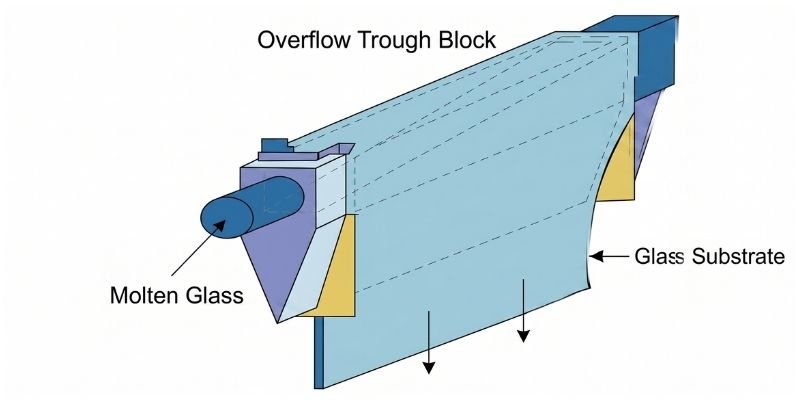
Forming Process Flow
The forming process involves:
- Glass melting
- Refining and homogenization through platinum channels
- Temperature reduction
- Overflow forming inside a muffle furnace
- Controlled cooling
- Annealing for stress removal
- Horizontal and vertical cutting
- Thickness inspection
- Application testing
- Electrostatic dust removal
- Final appearance inspection
Semi-finished substrate glass is then transferred to BOD (semi-finished processing) operations.

High-Generation and Ultra-Thin Development Trends
To match downstream panel manufacturing requirements, substrate glass production lines are classified by generation size based on glass area output.
Larger substrate sizes correspond to higher-generation production lines.
Currently, Gen 10 production lines are the largest in operation, with substrate dimensions reaching:
2880 × 3130 mm
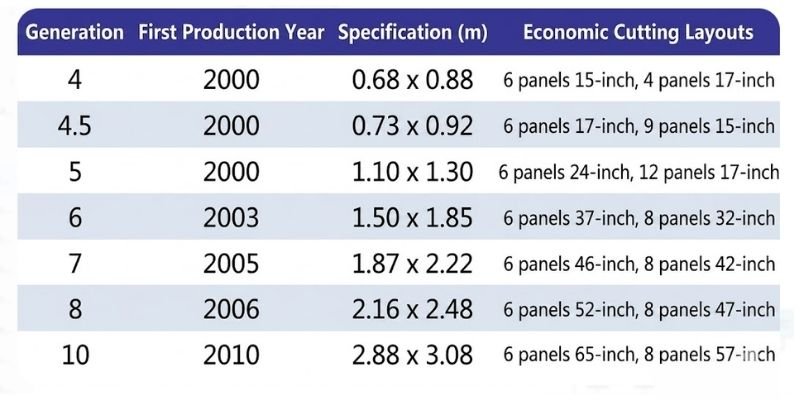
As panel manufacturers continue advancing toward larger-generation lines, substrate glass manufacturers are following the same trend. Major global glass manufacturers are now focusing primarily on high-generation production capacity expansion.
Three Major Technical Barriers in the Substrate Glass Industry
Although substrate glass may appear simple, the industry has extremely high technical barriers.
These barriers can be divided into three categories:
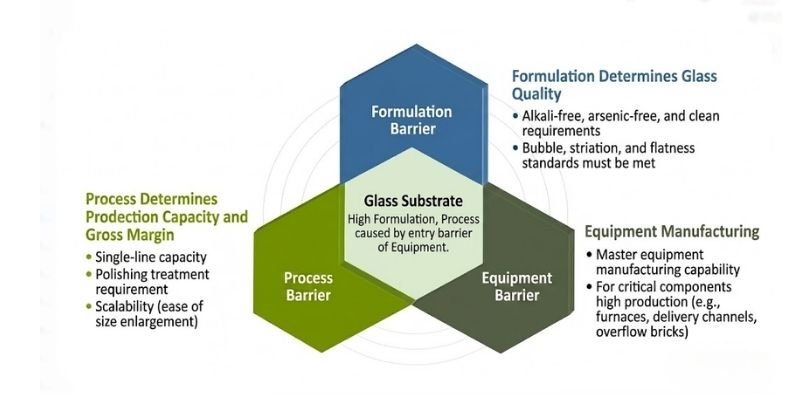
1. Process Technology Barrier
TFT-LCD substrate glass requires electronic-grade surface flatness and impurity control.
Conventional float processes struggle to meet these standards, while polishing may introduce additional contaminants.
Overflow fusion processing requires precise control of:
- Temperature
- Flow rate
- Forming conditions
This makes the process highly complex.
2. Glass Formula Barrier
Glass formulation represents the core competitive barrier of the industry.
The overflow fusion process requires highly specialized glass compositions to achieve stable forming and optimal optical and chemical properties.
Traditional formulas often used arsenic oxide for bubble removal, but arsenic-containing glass faces environmental restrictions in many regions.
Developing arsenic-free formulations significantly increases technical difficulty.
Ultimately, glass composition directly determines manufacturing yield and product quality.
3. Equipment Barrier
Most substrate glass production equipment is internally developed by manufacturers themselves.
New market entrants must independently design and build production systems, including:
- Furnaces
- Overflow troughs
- Platinum channels
- Overflow bricks
Precision requirements are extremely high, and even small equipment deviations can significantly impact yield rates.
Industry Entry and Exit Characteristics
While entry barriers in the substrate glass industry are extremely high, exit barriers are relatively low.
Compared with LCD panel production lines requiring investments of tens of billions of RMB, a Gen 6 substrate glass production line typically requires around RMB 700 million in investment.
Additionally, platinum equipment investments can often be partially recovered through resale.
Conversion from Substrate Glass to Cover Glass
Substrate glass manufacturers have both the capability and motivation to convert production lines to cover glass manufacturing.
Cover glass is the strengthened outer protective glass used in smartphones, tablets, and ultrabooks.
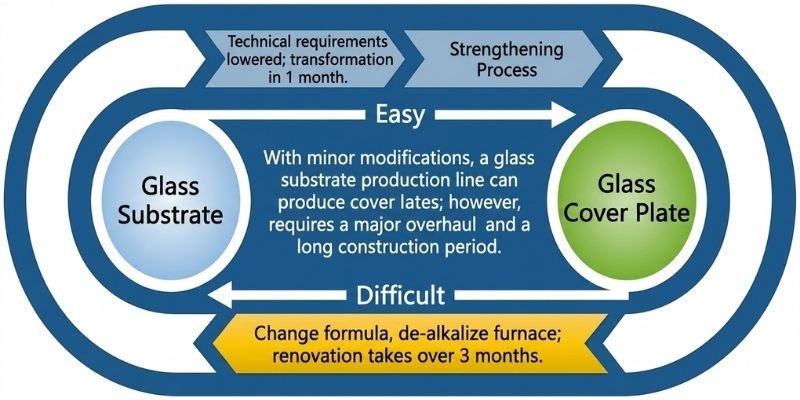
Compared with substrate glass:
- Surface flatness requirements are lower
- Alkali-free formulations are not mandatory
- Manufacturing difficulty is lower
Converting substrate glass lines into cover glass production lines is relatively straightforward and may take only about one month with proper formulations.
However, converting back to substrate glass production is significantly more difficult due to alkali contamination and higher thermal resistance requirements.
Corning’s “Gorilla Glass” Strategy
As smartphone and tablet demand surged, cover glass demand also increased rapidly.
Corning’s “Gorilla Glass” became widely adopted by major brands including Apple and became a preferred cover glass solution for flagship smartphones such as the iPhone.
Due to strong profitability in the cover glass market, Corning converted multiple substrate glass production lines into Gorilla Glass production lines between 2011 and 2012.
This demonstrated how substrate glass manufacturers can leverage advanced glass technologies to expand into other high-value specialty glass markets.

Major Global Cover Glass Manufacturers
Chinese manufacturers such as Dongxu and Rainbow Group have also introduced advanced aluminosilicate cover glass technologies using float and overflow fusion methods.
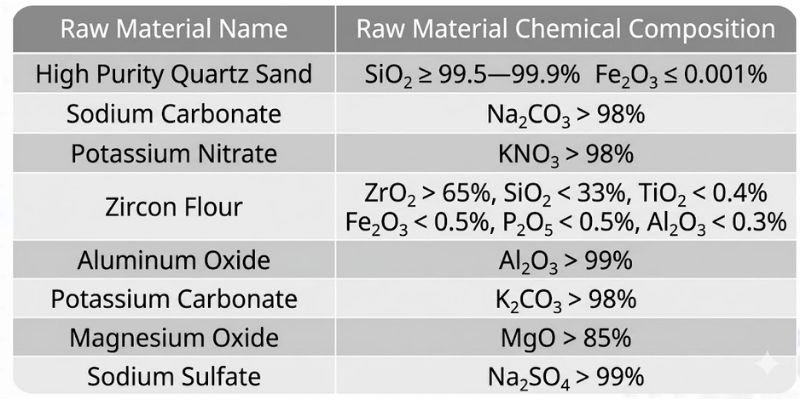
Main Raw Materials for High-Alumina Glass
The primary raw materials for high-alumina glass include:
Conclusion
Unlike capital-intensive panel manufacturing, substrate glass manufacturing is fundamentally a technology-driven industry.
High technical barriers have resulted in:
- Strong supply-demand dynamics
- Oligopolistic market structures
- High profit margins
Only companies capable of producing qualified products can participate in this highly profitable market.
For many years, the substrate glass industry has been dominated by leading manufacturers such as Corning and major Japanese glass companies due to the significant technological barriers involved.
Source: Compiled from GF Securities, Industry Information Network, Rainbow Group, Dongxu, and other industry sources.